
Fall leaves at Driftwood Lodge Resort, Lake Kabetogama. Toward the end of summer, the deep water becomes quite depleted of oxygen because no mixing has taken place. The middle layer, known as the thermocline, acts as an effective barrier to any mixing of the deeper waters. Because of the density-temperature relationship, many lakes in temperate climates tend to stratify, that is, they separate into distinct layers. Atmospheric oxygen is added by the air-water interaction to the oxygen produced within the water by aquatic plants.īut there is too much difference in temperature between the surface water and that at depth to allow for complete mixing of all the water in the lake. Winds and storms can cause some mixing and do add some oxygen. Starting in the spring and over the course of the summer, surface waters absorb a lot of the sun’s energy and can heat extensively, causing them to become quite buoyant. So how does all of this relate to lake turnover (which by the way, can actually take place once or several times per season, depending on many additional factors)? Let’s first consider the fall turnover. Photo by Yvonne Carpenter-Ross Lake Oxygen & the Thermocline Ice developing along the shores of Green Lake at the Heidel House Resort. All water beneath the surface of the ice never gets any colder than 39 degrees Fahrenheit. It also stops further energy loss from the lake. It dictates that at 39 degrees (39.20 to be precise) Fahrenheit, water actually expandsand becomes less dense, allowing it to float above the warmer water! The water that cools below that temperature, to 32 degrees, for instance, freezes and stays on the top, effectively capping the lake. Why then does this not happen? Again, it’s the chemistry of the water molecule. This would mean that the lake would freeze from the bottom up, eventually destroying all life beneath the waves. With continued cooling at the surface, the decreasing temperature should (or so it would seem) eventually cause all the water in the lake to eventually freeze solid and sink to the bottom. Ultimately sinking to the bottom of the lake, this cooler water pushes the water it has displaced back to the surface, where it, too, can cool. In any case, as water cools to that temperature, it does, as predicted, contract and becomes denser. Well, it is wonderful for lakes and the life in them, not so for the roads on which we drive. Something strange and wonderful happens when water reaches a temperature of approximately 39 degrees Fahrenheit (4 Celsius). Photo by Yvonne Carpenter-Ross Magic Happens at 39 Degrees Fahrenheit Fog on lake in St Germain, WI during morning of musky fishing tournament. Able to exist in all three forms, water, as a liquid, contracts and becomes denser as it gets colder… at least to a point.Īnd that point is where the oddity owing to water’s special chemistry comes into play, the one that keeps your favorite lake and your favorite gamefish, healthy and happy. 
This principle holds for all solids, liquids and gases. As they get warmer, they expand and become less dense/lighter. 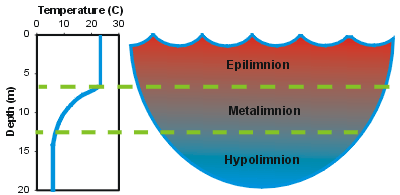
In general, as things get colder, they contract and become more dense/heavier. In nature, temperature changes the physical properties of all substances in predictable ways. One of the more interesting reasons is because of a water molecule’s amazing structure. Water is fascinating, for a number of reasons. Photo by Pixabay on Pexels Some Basic Chemistry: Hot & Cold But I’ll try to describe the process in a way that is as simple and non-technical as possible. Did I say simple? Well… it is, and then again it isn’t. In terms of humans, I recently heard of a new way to calculate how much water one should consume each day: Take one’s body weight in pounds and divide it by two, and that is the number of ounces of water required daily! I am unaware of the requirements for other warm-blooded animal forms, but I do know that the cold-blooded species so cherished by sports enthusiasts - fish like walleye, bass, northern pike and musky - require a lotof water: fresh, clean and oxygen-laden H2O in order to survive.Īnd a simple process referred to as lake turnover plays a very critical role in that survival process. This is true for a number of reasons, not the least of which is that without water, life as we know it would not exist. Water is quite possibly the most fascinating substance on earth. After some extensive research, here’s what I’ve learned. What is Lake Turnover? Does it effect fishing? What’s happening down there? I had pondering these questions for some time, especially after taking some fishing trips in the fall.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
